Synthetic access to activated iron compounds may replace precious metals as catalysts
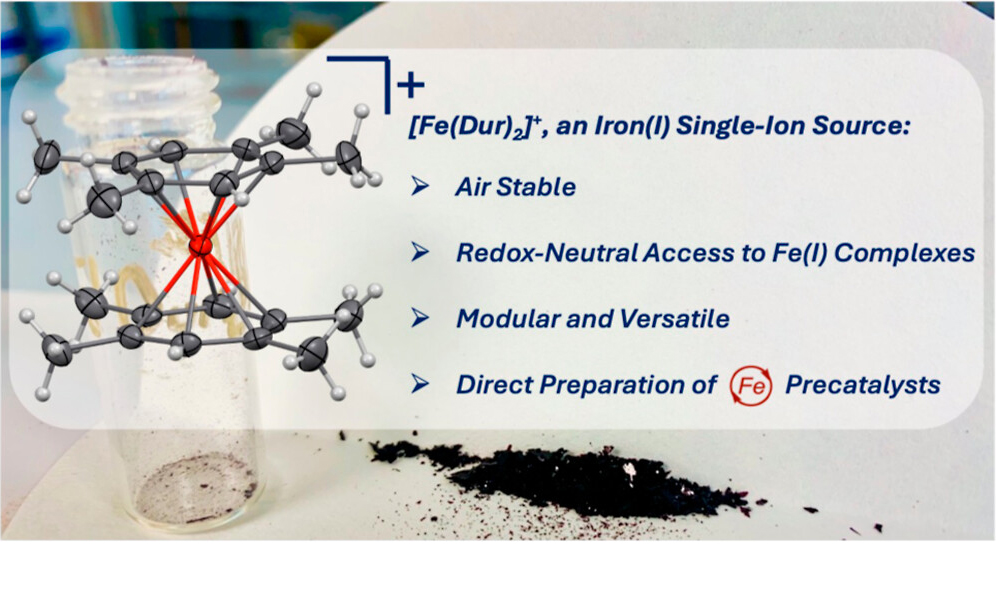
For the chemical industry, rare precious metals are essential as catalysts to accelerate chemical reactions. Iron compounds represent a sustainable alternative that is abundant in nature. In contrast to many precious metals, however, iron does not have an available starting material in its reactive state. Oliver Townrow and his group have now developed a mild synthetic method for the selective production of single positively charged iron complexes. Thus, they no longer depend on the use of strong reducing agents which are hazardous and require specialist handling. The new process enables them to directly produce a range of known and novel iron complexes that work as catalysts in industrially relevant transformations. The results are published in the Journal of the American Chemical Society.
“Our goal is to establish iron as a sustainable swap-in alternative for precious metal catalysis and to explore new applications, without changing the logistics and techniques used for the current state-of-the-art”, says principal investigator Dr. Oliver Townrow. “We have already shown that these compounds, which are simply comprised of iron bonded to inexpensive aromatic hydrocarbons, are remarkably stable in air in the solid state making them attractive for industrial use.” With his KIT junior research group Functional Organometallic Chemistry, Townrow conducts research at the Institute of Nanotechnology at KIT.